energy and place
How does energy production and consumption impact place?
Project Reflection
-What did you learn about energy or energy resources from your particular scientific investigation?
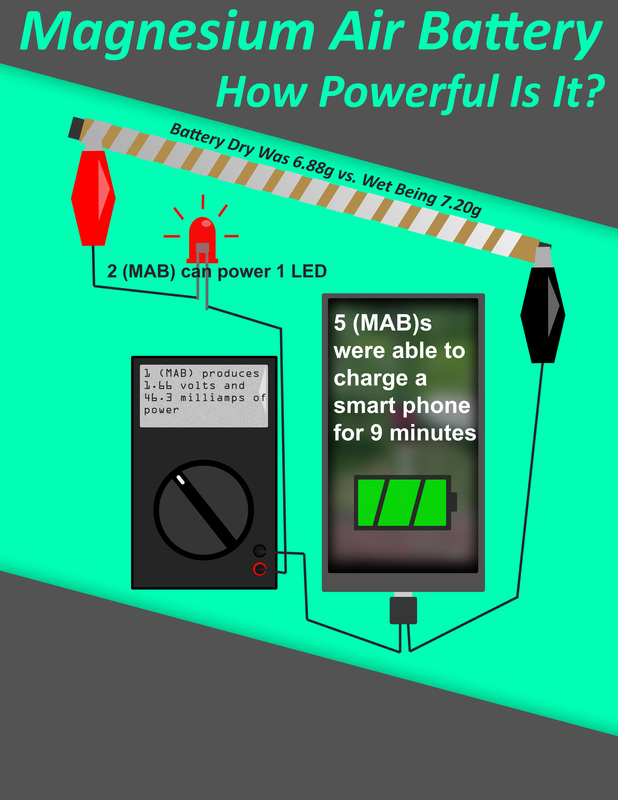
I realized how hard it is to to produce the energy we use everyday. Even the battery my group made did not produce much power. The amount of energy that is required to keep modern society going is an amazing feat of modern engineering that we can keep up with such demand. Batteries are the biggest bottleneck in renewable resources because we can't store the power from solar and wind long enough. That is why finding better batteries will revolutionize the renewable energy movement.
-What did you learn about the nature of science by researching, designing, conducting, analyzing and writing
about your own investigation? How has this shaped your perspective about science and scientists?
We had to research our idea and find out how to make our batteries. Then we had to test outer batteries while also discovering we could charge a phone. Then we but all of the data we collected into a scientific paper. It was interesting to work collaboratively with my in a group just like professional scientist would allowing our group to play off each other's strengths allowing us to produce better work. Science is a challenging field and I have the up most respect for the hard work that scientists do.
-What did you learn about energy or energy resources from your particular scientific investigation?
I realized how hard it is to to produce the energy we use everyday. Even the battery my group made did not produce much power. The amount of energy that is required to keep modern society going is an amazing feat of modern engineering that we can keep up with such demand. Batteries are the biggest bottleneck in renewable resources because we can't store the power from solar and wind long enough. That is why finding better batteries will revolutionize the renewable energy movement.
-What did you learn about the nature of science by researching, designing, conducting, analyzing and writing
about your own investigation? How has this shaped your perspective about science and scientists?
We had to research our idea and find out how to make our batteries. Then we had to test outer batteries while also discovering we could charge a phone. Then we but all of the data we collected into a scientific paper. It was interesting to work collaboratively with my in a group just like professional scientist would allowing our group to play off each other's strengths allowing us to produce better work. Science is a challenging field and I have the up most respect for the hard work that scientists do.
History of the Silicon Transistor
Technology is everywhere. We have smartphones, laptops, iPads, and now even smart watches are starting to show up. The question is what makes them tick. These almost magical machines need one abundant element called silicon. Silicon is a non-conductive metalloid that is just below carbon on the periodic table. Raw silicon is unusable for electronics because it is an insulator, that is, electricity will not pass through it. However, through multiple chemical processes called "doping" where elements like Indium or Gallium are used because of the fact that they have the one more or one less valence electron and they also have a very similar atomic radius. These elements are used to add conductive lines called traces that act like extremely small wires, silicon can act as an insulated conductor. This process turns the silicon into a semiconductor capable of being conductive or nonconductive based on whether a voltage is applied. The resulting device serves as an on-off switch controlled by electricity, creating the transistor. This happens because the transistor is split up into two parts known as n-type and p-type; the standard configuration for this is (N-P-N). The p-type is positive because it was doped with an element like boron and the n-type is doped with an element like arsenic. When applied to the silicon in the N-P-N configuration the gap between the p and the n type becomes something called a depletion layer where electrons from the n-type move over to the p-type creating a negatively charged barrier. This barrier repels the elections, but when a positive charge is applied it pulls the election with enough force to overcome the barrier letting the elections through. When many of these transistors are chained together they create an integrated circuit that is at the heart of modern devices.
You may be wondering how such a discovery like the silicon transistor came about. It was first created by Morris Tanenbaum in January 1954 at Bell Labs using existing grown-junction techniques. The silicon transistor was created to replace the transistors made out of germanium which has a very low temperature tolerance; 70℃ would cause the transistors to fail. But Bell Labs did not research it further because they didn't believe that it was marketable for commercial use. The company Texas Instruments later continued working on the silicon transistor. Chemist Willis Adcock lead a team to make their first silicon transistor using high grade silicon. Shortly thereafter the silicon transistor was available for sale, allowing TI to dominate the silicon transistor market for many years. By 1950s, the silicon transistor became the preferred transistor.
In 1952, Bell Labs chemist Calvin Fuller came up with a way to introduce impurities into germanium by exposing the germanium to extremely hot gases with elements (called dopants) in them. He was able to control the impurities by controlling variables like time and temperature. Calvin Fuller was now able to control the depth of the impurities with accuracies of better than one micrometer. This discovery made Fuller’s work with engineer Daryl Chapin and physicist Gerald Pearson possible. In 1954 Calvin Fuller and his team diffused boron atoms into wafers of silicon the resulting in junctions below the wafer’s surface. The resulting junctions would generate voltage when light was shined on them through the photovoltaic effect and how the charges move through the silicon. This discovery would later be known as the solar panel. Later in 1954 the same diffusion process would be used to create a silicon transistor that could run at 170 MHz or ten times higher than previous silicon transistors meaning that it turns off and on many times a second.
The silicon transistor has become an integral part of society allowing for the technological leaps that we see today. The science behind the silicon transistor has led to discoveries like the solar panel. The silicon transistor has led to an industrial revolution where computers and devices are getting smaller, better, and faster. We owe modern day life to the pioneers of the silicon transistor, like Calvin Fuller, who came up with the process that made this technology possible.
You may be wondering how such a discovery like the silicon transistor came about. It was first created by Morris Tanenbaum in January 1954 at Bell Labs using existing grown-junction techniques. The silicon transistor was created to replace the transistors made out of germanium which has a very low temperature tolerance; 70℃ would cause the transistors to fail. But Bell Labs did not research it further because they didn't believe that it was marketable for commercial use. The company Texas Instruments later continued working on the silicon transistor. Chemist Willis Adcock lead a team to make their first silicon transistor using high grade silicon. Shortly thereafter the silicon transistor was available for sale, allowing TI to dominate the silicon transistor market for many years. By 1950s, the silicon transistor became the preferred transistor.
In 1952, Bell Labs chemist Calvin Fuller came up with a way to introduce impurities into germanium by exposing the germanium to extremely hot gases with elements (called dopants) in them. He was able to control the impurities by controlling variables like time and temperature. Calvin Fuller was now able to control the depth of the impurities with accuracies of better than one micrometer. This discovery made Fuller’s work with engineer Daryl Chapin and physicist Gerald Pearson possible. In 1954 Calvin Fuller and his team diffused boron atoms into wafers of silicon the resulting in junctions below the wafer’s surface. The resulting junctions would generate voltage when light was shined on them through the photovoltaic effect and how the charges move through the silicon. This discovery would later be known as the solar panel. Later in 1954 the same diffusion process would be used to create a silicon transistor that could run at 170 MHz or ten times higher than previous silicon transistors meaning that it turns off and on many times a second.
The silicon transistor has become an integral part of society allowing for the technological leaps that we see today. The science behind the silicon transistor has led to discoveries like the solar panel. The silicon transistor has led to an industrial revolution where computers and devices are getting smaller, better, and faster. We owe modern day life to the pioneers of the silicon transistor, like Calvin Fuller, who came up with the process that made this technology possible.
LINKS
http://www.computerhistory.org/semiconductor/timeline/1954-Silicon.html
How Does a Transistor Work?: http://youtu.be/IcrBqCFLHIY
http://www.computerhistory.org/semiconductor/timeline/1954-Silicon.html
How Does a Transistor Work?: http://youtu.be/IcrBqCFLHIY
Reflection
“How has the chemistry of materials shaped our past, present and how may it shape our
future?”
Chemistry has changed our past in so many ways, many of which have changed how life is today. One very good example is the silicon transistor that i rote about in the writing above. if it wasn't for the silicon transistor you wold not be reading this right now, if it wasn't for the vulcanization of rubber we probably wouldn't be using rubber for tires. Chemistry will also play a huge role in our futures, materials like carbon nano types and graphene will shape the future and make the world a better place for all of us. Materials and chemistry have shaped the world past, present, and future and as the saying goes “better living through chemistry”.
“How does the structure of matter on the atomic, molecular, microscopic and macroscopic
levels determine a material’s properties?”
Based on how the atoms in the material are bonded changes how it behaves like in a carbon nanotube. In a carbon nanotube the carbon is bonded three times letting it share three elections with other carbon atoms. Carbon needs 4 elections to have a full valence shell and with three being shared with other carbon in the nanotube it only needs one more election to fulfill the octet rule. Because of this gap carbon nanotubes are very conductive, this is due to elections "hopping" from gap to gap. Other materials also gain their properties from how they are bonded, like polymers that tend to be long chains of atoms and are generally quite strong. These are just a few examples of how the structure of a materials atoms changes how it behaves.
“How has the chemistry of materials shaped our past, present and how may it shape our
future?”
Chemistry has changed our past in so many ways, many of which have changed how life is today. One very good example is the silicon transistor that i rote about in the writing above. if it wasn't for the silicon transistor you wold not be reading this right now, if it wasn't for the vulcanization of rubber we probably wouldn't be using rubber for tires. Chemistry will also play a huge role in our futures, materials like carbon nano types and graphene will shape the future and make the world a better place for all of us. Materials and chemistry have shaped the world past, present, and future and as the saying goes “better living through chemistry”.
“How does the structure of matter on the atomic, molecular, microscopic and macroscopic
levels determine a material’s properties?”
Based on how the atoms in the material are bonded changes how it behaves like in a carbon nanotube. In a carbon nanotube the carbon is bonded three times letting it share three elections with other carbon atoms. Carbon needs 4 elections to have a full valence shell and with three being shared with other carbon in the nanotube it only needs one more election to fulfill the octet rule. Because of this gap carbon nanotubes are very conductive, this is due to elections "hopping" from gap to gap. Other materials also gain their properties from how they are bonded, like polymers that tend to be long chains of atoms and are generally quite strong. These are just a few examples of how the structure of a materials atoms changes how it behaves.